What is this unusual fuel called "Steam-reformed Hexane"?
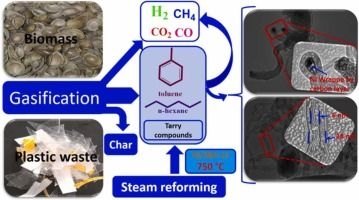
When you hear the topic "Steam-reformed hexane," it might sound a bit complicated. However, simply put, it refers to the process of subjecting a hydrocarbon liquid called Hexane to a chemical reaction with steam to produce a "Hydrogen-rich" fuel. This is an incredibly important topic in both the industrial world and the automotive sector. Especially as we discuss the fuels of the future, understanding this technology is highly valuable for a vehicle owner like you.
What is Hexane (C₆H₁₄)?
First, let's look at what Hexane (C₆H₁₄) actually is. It is a major component found in petrol (gasoline). It is commonly used to increase the octane rating of standard petrol and also serves as an industrial solvent. However, instead of directly burning this hexane, "Reforming" it means altering its molecular structure to create a cleaner and more efficient energy source. This is achieved by using "Steam" (water vapor).
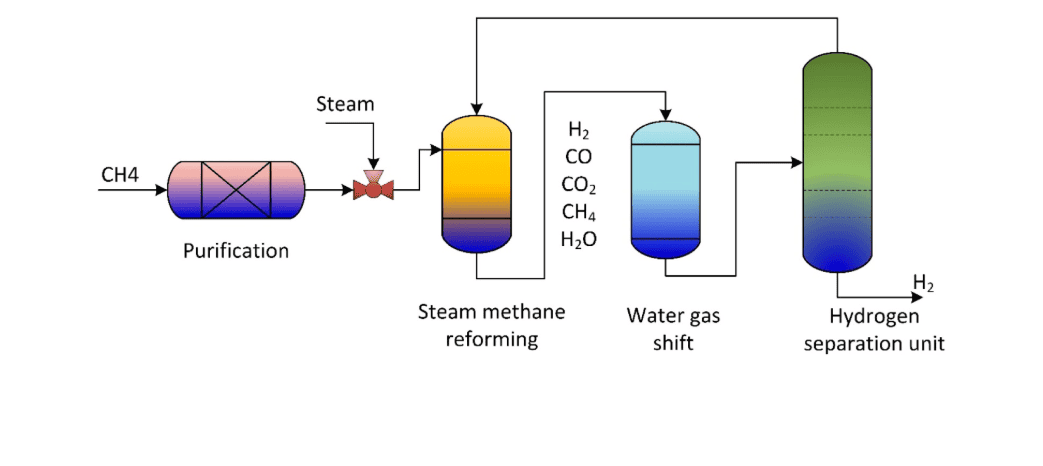
The Steam Reforming Process
Now let's talk about the industrial mechanism, known as the Steam Reforming Process. Think of this like a chemical recipe. We combine high-temperature hexane vapor with extremely high-temperature water vapor (Steam) and pass it over a catalyst, such as "Nickel." When this happens, the bonds between the carbon and hydrogen atoms in the hexane are broken, and they react with the oxygen present in the water vapor.
The primary results of this reaction are Hydrogen gas (H₂) and Carbon Monoxide (CO). In the industrial sector, this mixture is called "Syngas" (Synthetic Gas). This Syngas can then be further purified to extract pure hydrogen. As we know, hydrogen is the ultimate fuel for future "Zero Emission" vehicles. Therefore, being able to produce hydrogen from a liquid fuel like hexane is a massive advantage in terms of transportation.
Why is this Process So Important?
Usually, storing and transporting hydrogen gas is very difficult because it is a highly volatile gas. However, hexane is a stable liquid at room temperature. Because of this, we can fill a vehicle's tank with liquid hexane, use this "On-board reforming" technology inside the vehicle itself, and instantly produce the hydrogen needed to run the engine or feed a Fuel Cell. It is essentially like carrying a mobile hydrogen factory inside your car.
Technical Challenges: Thermal Efficiency and Carbon
Looking at it from a technical perspective, we have to pay close attention to Thermal Efficiency. To reform hexane, a very high temperature of around 700°C to 1000°C is required. To generate this heat, a certain amount of fuel must be burned. Therefore, the overall efficiency of this entire process depends entirely on how well we manage that heat.
Another factor is the carbon byproducts emitted during this process. Because hexane is a carbon-based fuel, Carbon Dioxide (CO₂) is emitted. However, because this reforming process can be done much more efficiently and in a highly controlled manner compared to burning petrol in a traditional internal combustion engine, it is considered a strong alternative to minimize pollution. It is crucial to understand that this is not just "burning" fuel; it is a controlled chemical conversion.
The Future of the Technology
Currently, this steam-reformed hexane technology is mostly used in large industrial plants to produce hydrogen. However, at the research level, massive efforts are underway to scale this down to smaller, mobile sizes. For example, the automotive world is heavily focused on systems where hydrogen-powered buses or trucks can instantly generate their own hydrogen on the go using hexane or similar liquid hydrocarbons.
Conclusion
Finally, this is not some bizarre, alien fuel; it is an advanced technological method of converting the energy locked inside an existing liquid fuel into much cleaner hydrogen energy. As a motorist, studying modern technologies like this greatly expands your knowledge. Remember, in the future, instead of simply burning oil, we will be extracting maximum energy through advanced chemical processes like these.

எழுதியவர்
Nisaga Sandaru
A versatile and highly experienced professional, currently an Automobile undergraduate at the University of Jaffna, with strong technical expertise grounded in hands-on work as a technician with SriLankan Airlines Ground Support Equipment (GSE). Holding an Automobile NVQ Level 3 qualification, along with specialized certifications in Vehicle ECU Programming and Diagnosis, and extensive experience in arc welding and vehicle body painting, and well equipped to handle complex automotive, diagnostic, and fabrication-related tasks with a high level of competence.









































